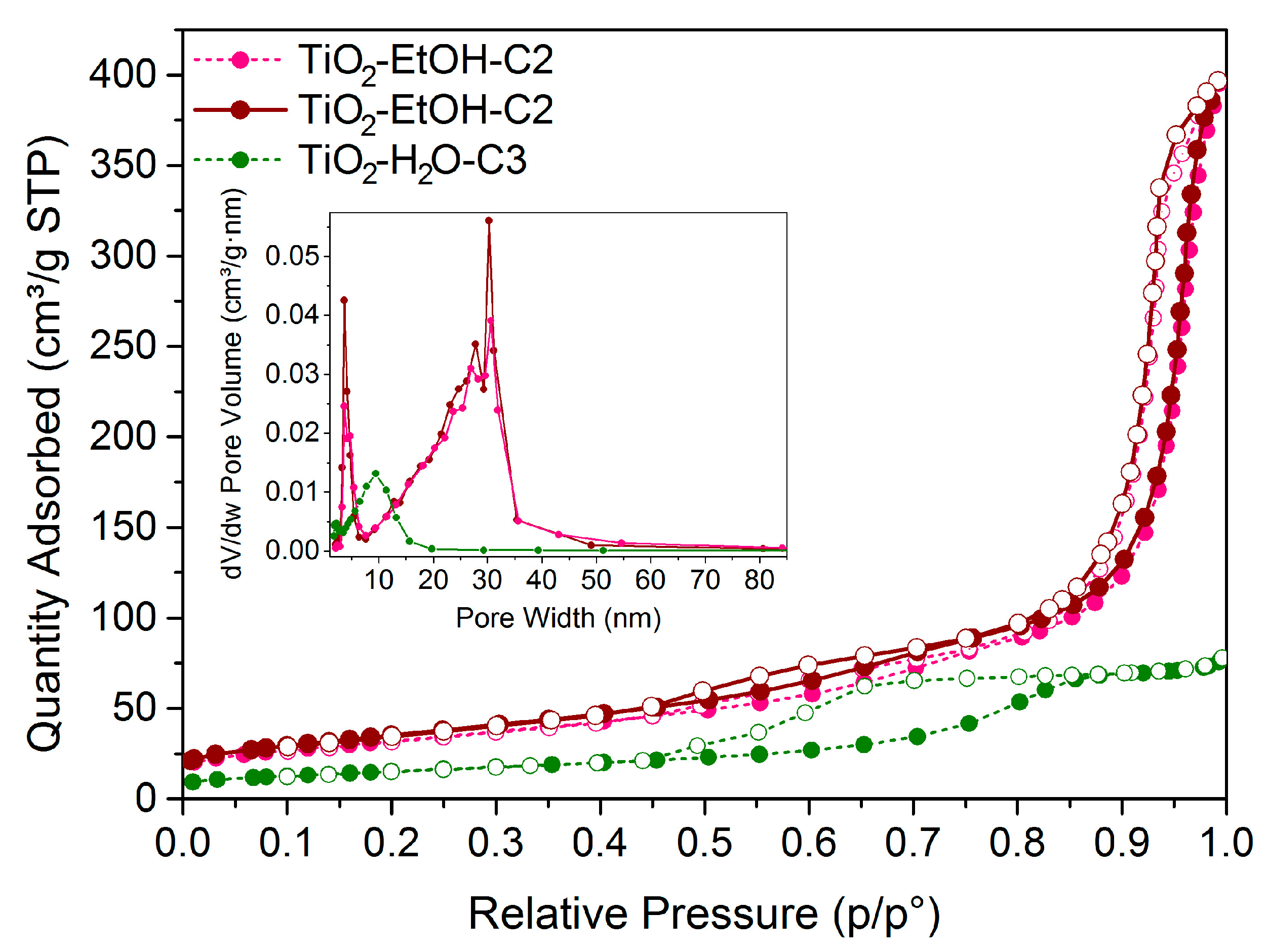
SOLVED: The reaction of 1.23 g TiCl4 (FW 189.71) with excess H2O according to the following reaction yields 0.456 g of TiO2 (FW 79.87). What is the percent yield? TiCl4 + 2
In situ ATR-FTIR study of H2O and D2O adsorption on TiO2 under UV irradiation - Physical Chemistry Chemical Physics (RSC Publishing)

Synthesis of Core‐Shell Au@TiO2@C Nanoparticles and Their Photocatalytic Properties for the Degradation of Rhodamine B Under Simulated‐Solar Light - Wang - 2020 - ChemistrySelect - Wiley Online Library

The Effect of SO2 and H2O on the Interaction Between Pt and TiO2(P-25) During Catalytic CO Oxidation | Catalysis Letters

Visualizing H2O molecules reacting at TiO2 active sites with transmission electron microscopy | Science

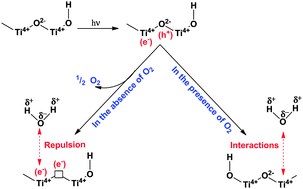
Basic processes on the surface of TiO2 in the presence of H2O and O2... | Download Scientific Diagram

a) The adsorption energy calculation of H2O on the TiO2 surface and H2O... | Download Scientific Diagram
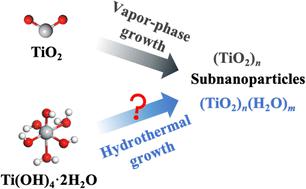
Aqueous growth of titania subnanoparticles: an understanding of the ultrasmall visible-light-absorbing unit of (TiO2)8(H2O)16,Physical Chemistry Chemical Physics - X-MOL
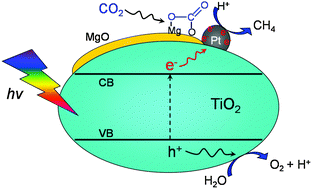
Photocatalytic reduction of CO2 with H2O: significant enhancement of the activity of Pt–TiO2 in CH4 formation by addition of MgO - Chemical Communications (RSC Publishing)

Full article: Effectively inhibiting particles aggregation and sedimentation for TiO2-H2O suspension by application of an electrode
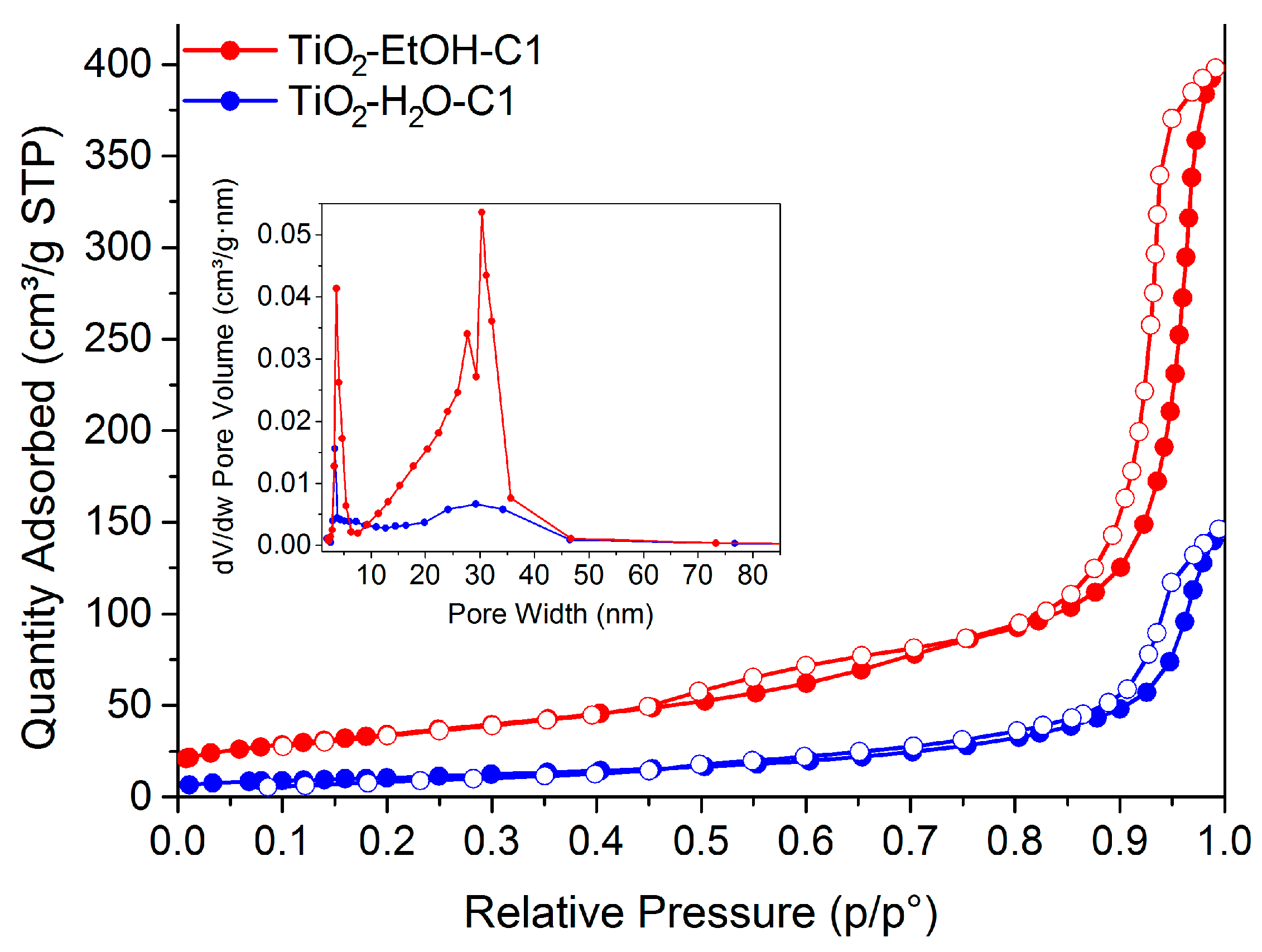
Nanomaterials | Free Full-Text | Novel Sol-Gel Synthesis of TiO2 Spherical Porous Nanoparticles Assemblies with Photocatalytic Activity

Computational analysis of comparative heat transfer enhancement in Ag-H2O, TiO2-H2O and Ag-TiO2-H2O: Finite difference scheme - ScienceDirect

TiO2 Phase Engineering by Millisecond Range Annealing for Highly Efficient Photocatalysis | The Journal of Physical Chemistry C

Water adsorption on TiO2 surfaces probed by soft X-ray spectroscopies: bulk materials vs. isolated nanoparticles | Scientific Reports