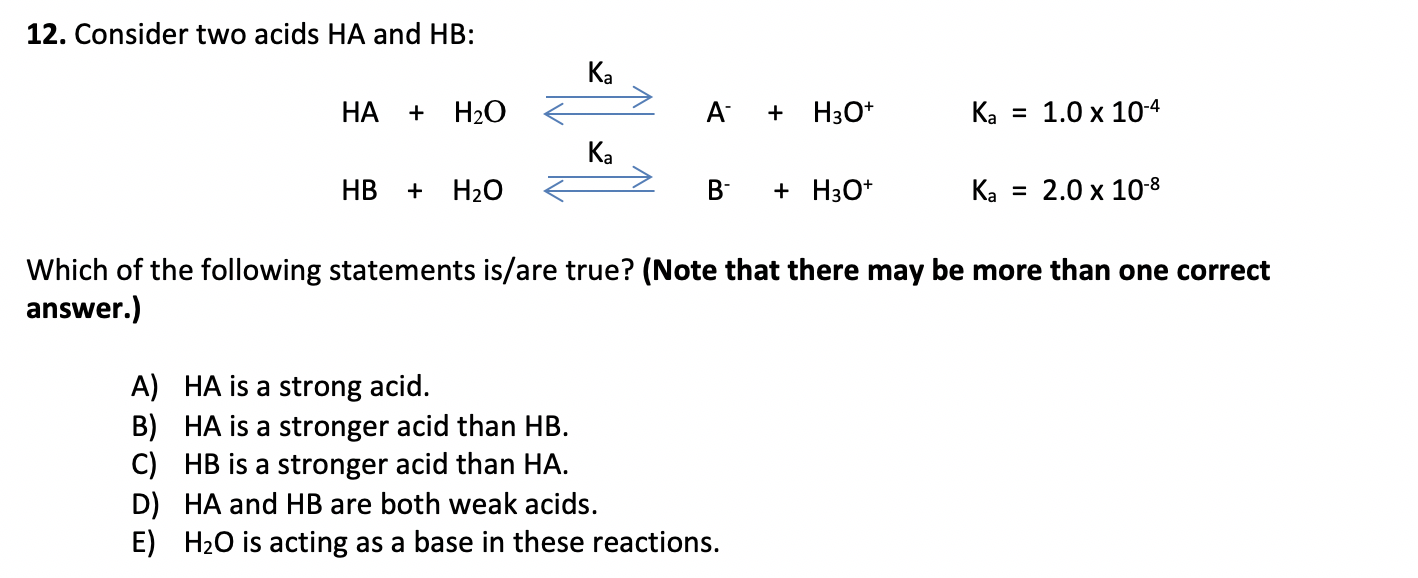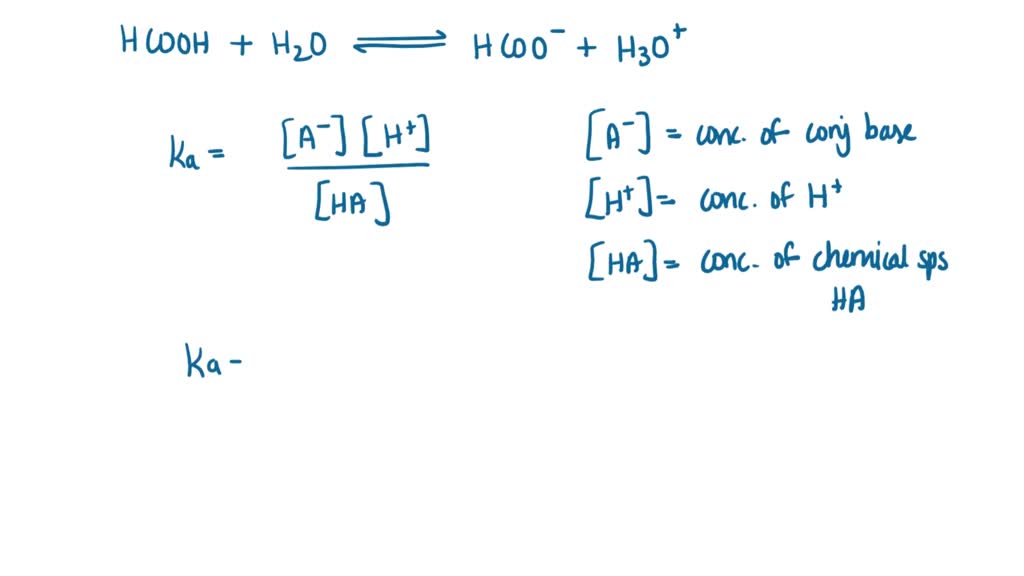
SOLVED: Write the acid dissociation constant expression, Ka, for the reaction below. HCO2H + H2O ⟷ HCO2- + H3O+

How to balance K+H2O=KOH+H2|Chemical equation K+H2O=KOH+H2|reaction balance K+H2O=KOH+H2| K+H2O= - YouTube

Oxidation Number method. K+H2O=KOH+H2. Balance the chemical equation by oxidation Number method. - YouTube

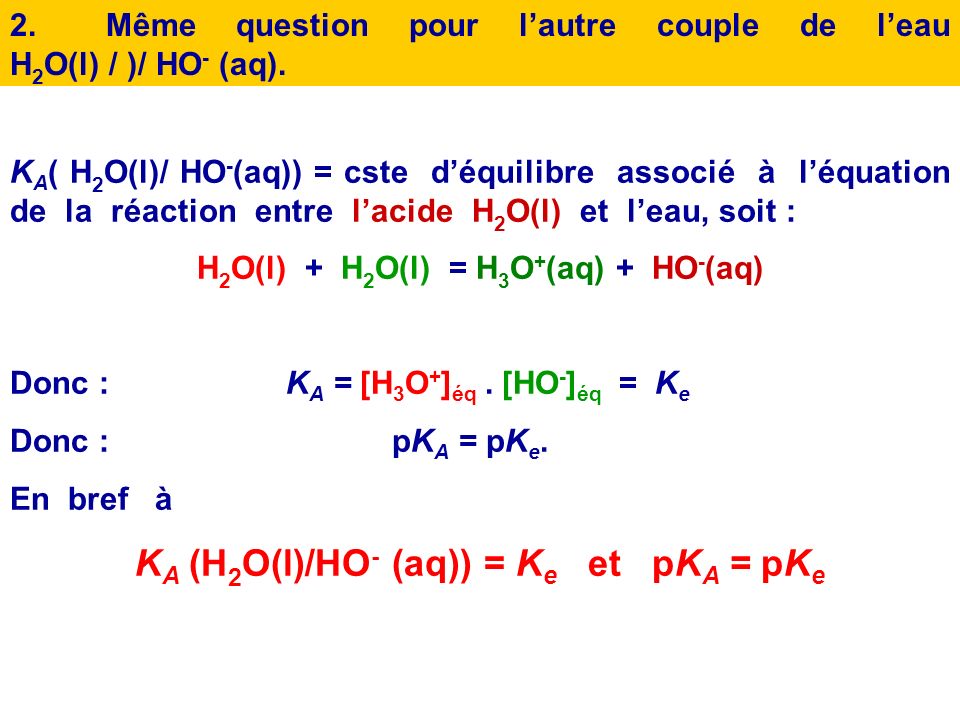
Water-exchange rate constants, k H2O , for a particular water molecule... | Download Scientific Diagram
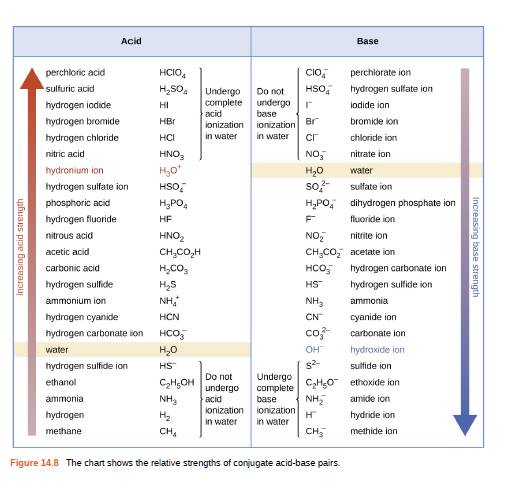
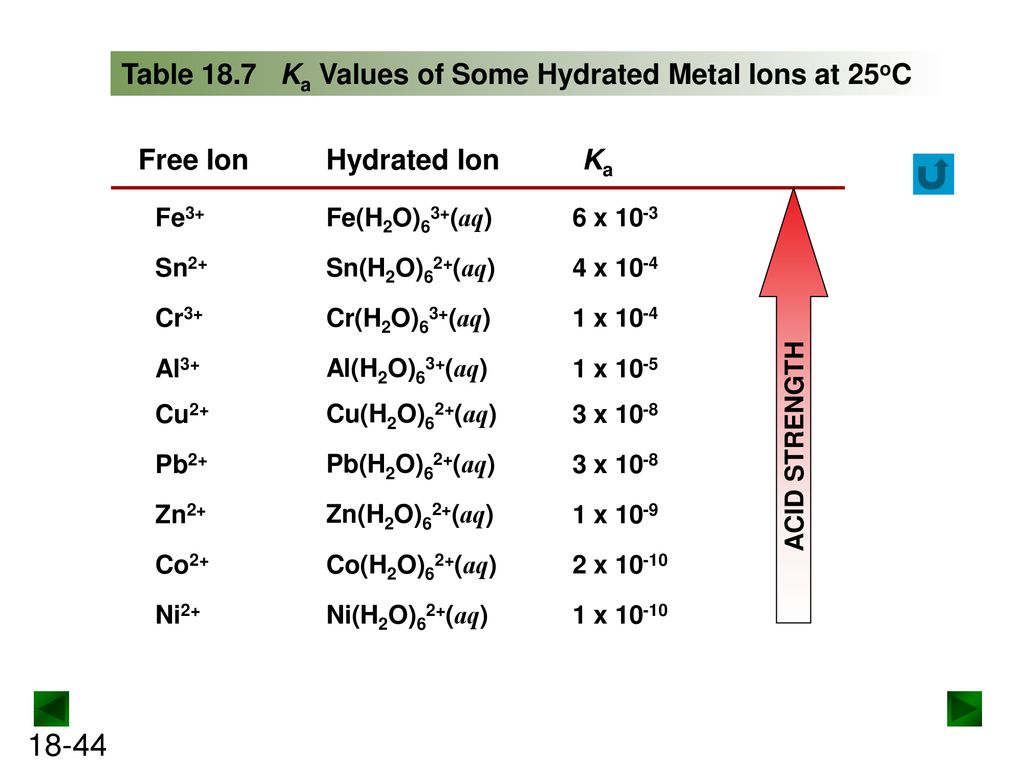
Using the K a value of 1.4 × 10 − 5 , place Al(H 2 O) 6 3 + in the correct location in Figure 14.8. | bartleby
SOLVED: The equilibrium constant Ka for the reaction Co(H2O)6^3+ (aq) + H2O (l) —> Co(H2O)5(OH)2+ (aq) + H3O+ (aq) is 1.0x10^-5 A) Calculate the pH of a 0.16 M solution of Co(H2O)6^3+.