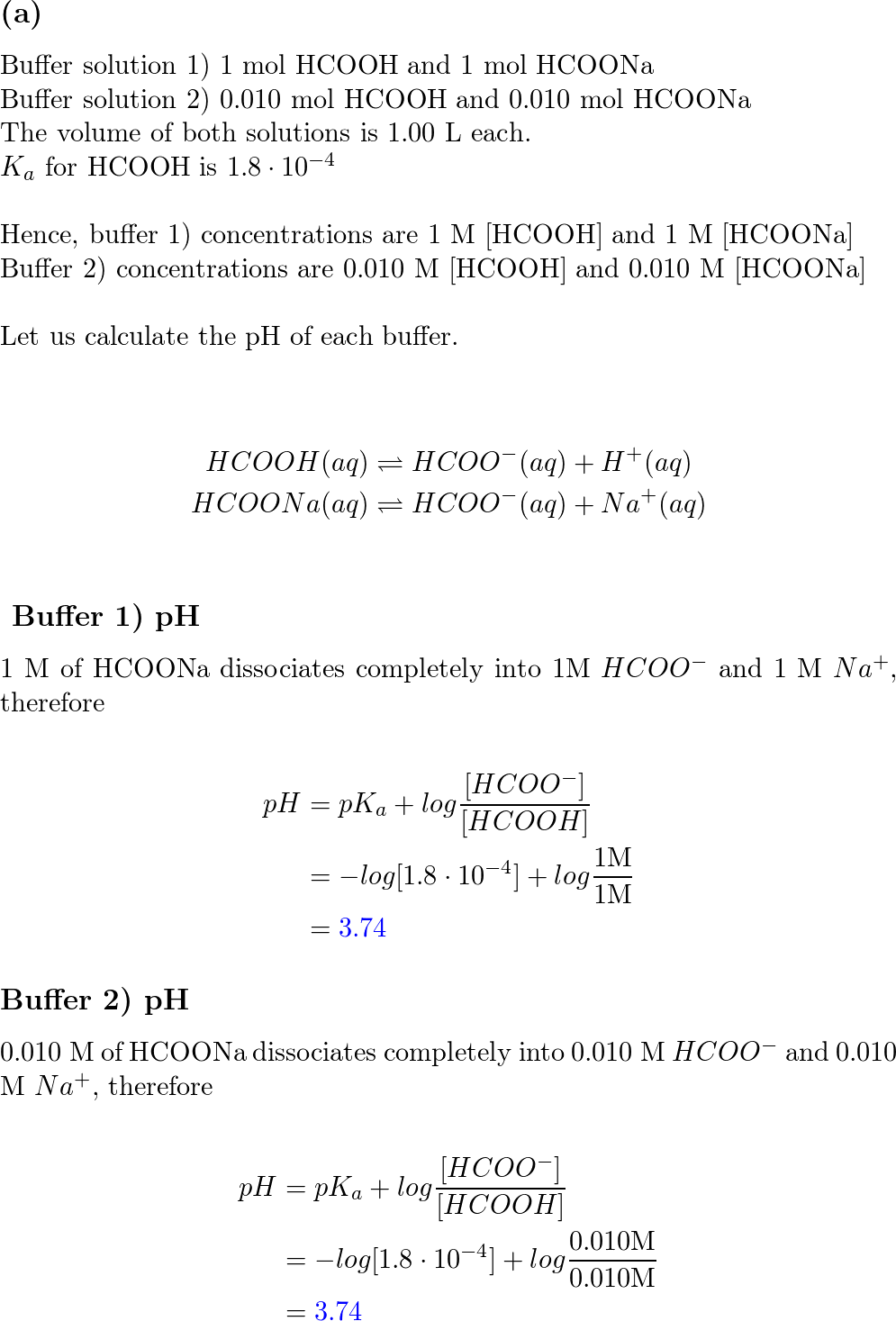
SOLVED: Which equation is correct for a buffer solution of formic acid and sodium formate? HCOOH(aq) + H2O(l) ⇌ H3O+(aq) + HCOO-(aq) HCOOH(aq) + NaOH(aq) ⇌ HCOONa(aq) + H2O(l) HCOOH(aq) + HCOONa(aq)

Experimental phase equilibrium data for the NPG (1) + HCOONa (2) +H2O... | Download Scientific Diagram

Find the equilibrium constant equilibrium HCOO + H2O HCOOH + OH- In a solution of 0.1 M HCOONa. Ka(HCOOH) = 1.8 * 104 (1) 1.8 x 10-4 (2) 5.56 x 10° (4) 1.8 * 10-18 (3) 5.56 x 107-11

Reductive Hydrodehalogenation of Halogenated Carboxylic Acid Derivatives Using a DMSO/HCOONa·2H2O System | Organic Letters

The aqueous solutions of HCOONa, C_(6)H_(5)NH_(3)CI, and KCN are, respectively, | 12 | IONIC EQ... - YouTube
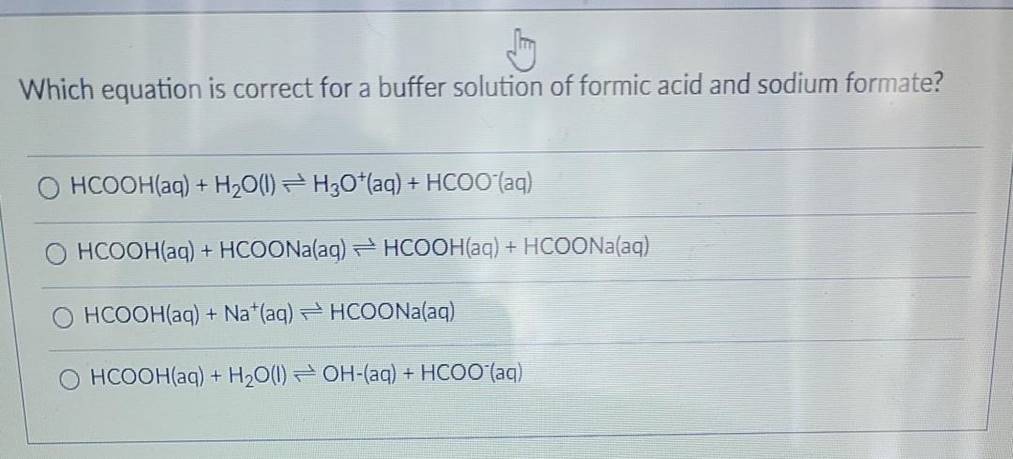
Solved) - Which equation is correct for a buffer solution of formic acid and... (1 Answer) | Transtutors

Find the equilibrium constant equilibrium HCOO + H2O HCOOH + OH- In a solution of 0.1 M HCOONa. Ka(HCOOH) = 1.8 * 104 (1) 1.8 x 10-4 (2) 5.56 x 10° (4) 1.8 * 10-18 (3) 5.56 x 107-11
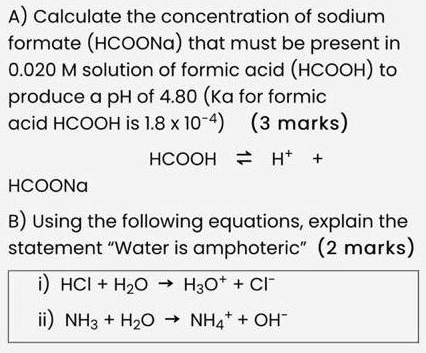
SOLVED: A) Calculate the concentration of sodium formate (HCOONa) that must be present in a 0.020 M solution of formic acid (HCOOH) to produce a pH of 4.80 (Ka for formic acid