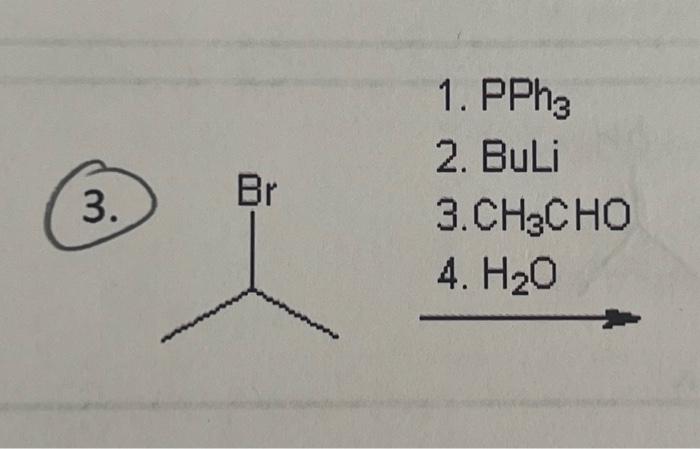
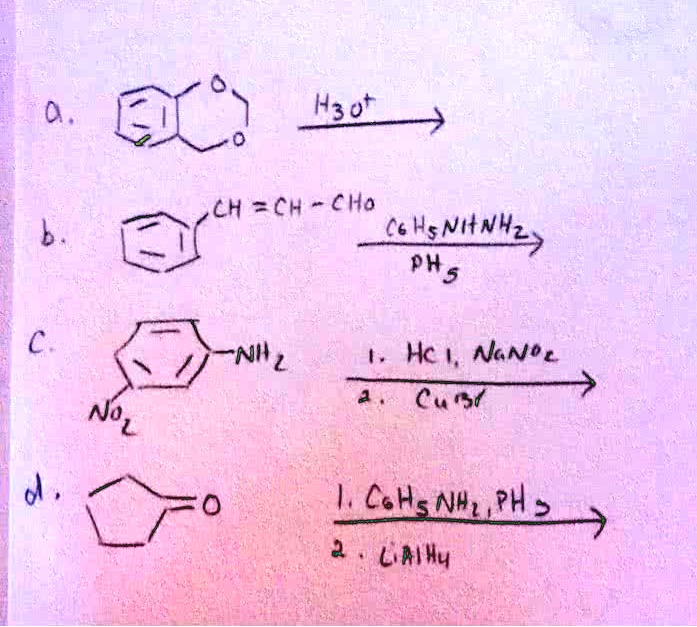
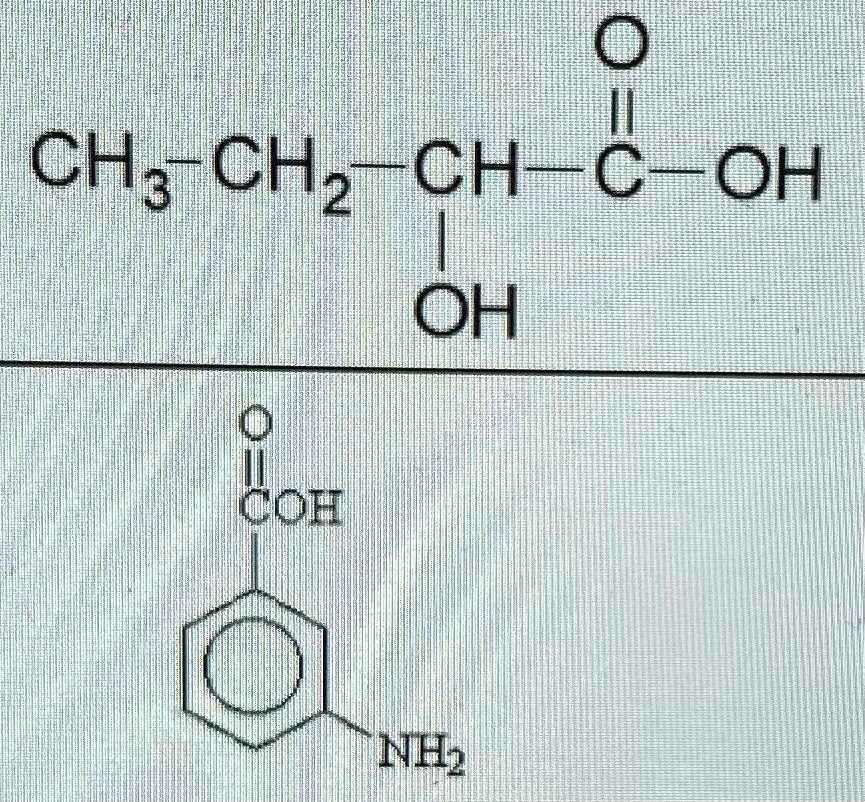
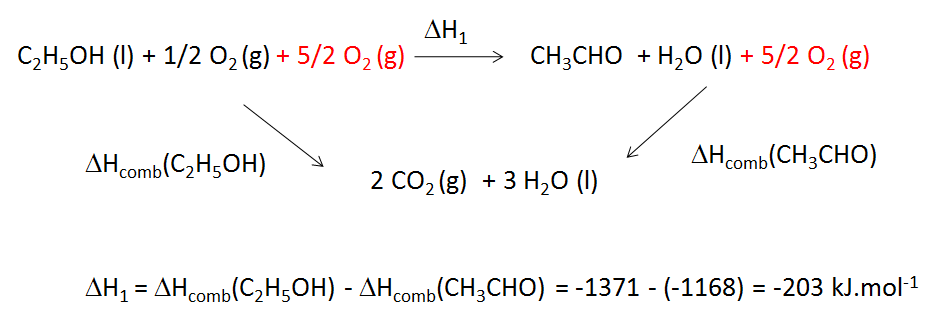
The concentration of CH3CHO increases from 0.0300 mol/L to 0.0500 mol/L in 42.5 seconds in the following - Brainly.in
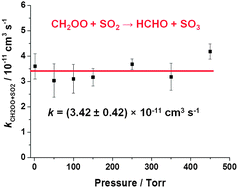
Kinetics of CH2OO reactions with SO2, NO2, NO, H2O and CH3CHO as a function of pressure - Physical Chemistry Chemical Physics (RSC Publishing)
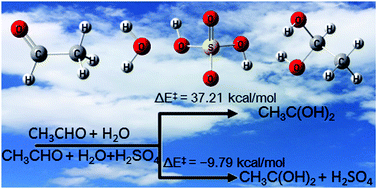
Atmospheric chemistry of CH3CHO: the hydrolysis of CH3CHO catalyzed by H2SO4 - Physical Chemistry Chemical Physics (RSC Publishing)

Atmospheric Formation of OH Radicals and H2O2 from Alkene Ozonolysis under Humid Conditions - Anglada - 2002 - ChemPhysChem - Wiley Online Library

A Sustainable Method for the Synthesis of Acetic Acid Based on Dehydrogenation of an Ethanol–Water Solution Catalyzed by an Iridium Complex Bearing a Functional Bipyridonate Ligand - Kuwahara - 2018 - ChemCatChem -
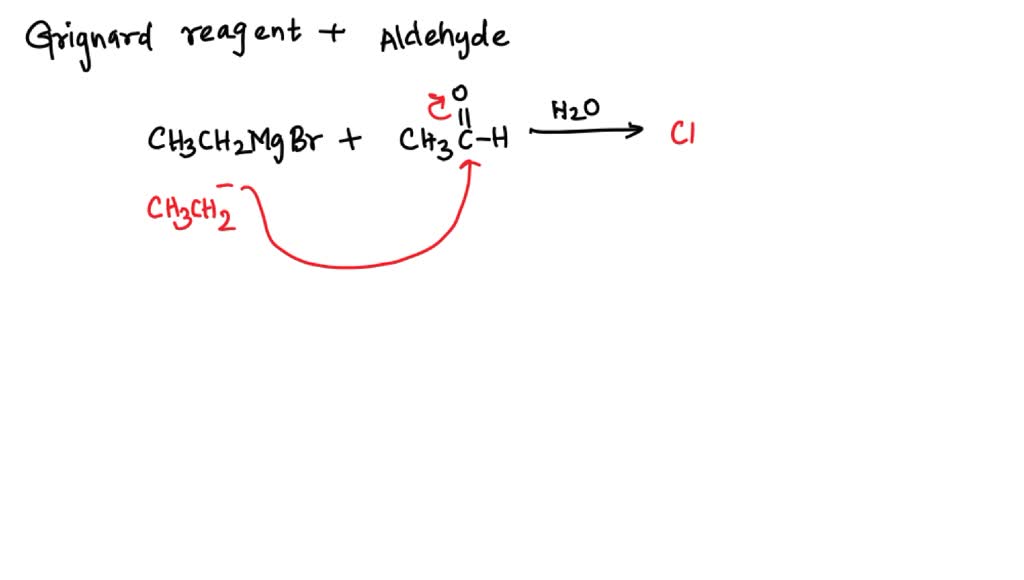
SOLVED: Text: CH3CH2MgBr CH3CHO/H2O(A) HBr(B) Alc.KOH(C) A. A, CH3CH2CHOHCH3; B, CH3 = CH2CHBrCH3; C, CH3CH = CH - CH3

SOLVED: 13.14 Acetaldehyde is produced from ethanol by the following gas-phase reactions: CH3CH2OH + O2 = CH3CHO + H2O CH3CH2OH = CH3CHO + H2 The reactions are carried out at 540°C and

Selective electrooxidation of acetaldehyde in aqueous ethanol alkaline solutions on silver-containing electrodes - ScienceDirect
Identify the reactant in the following reaction : (a) H2O (b) HCHO (c) CO (d) CH3CHO - Sarthaks eConnect | Largest Online Education Community

a Write the products formed when CH 3 CHO reacts with the following reagents :i HCNii H 2 N OHiii CH 3 CHO in the presence of dilute NaOHb Give simple chemical