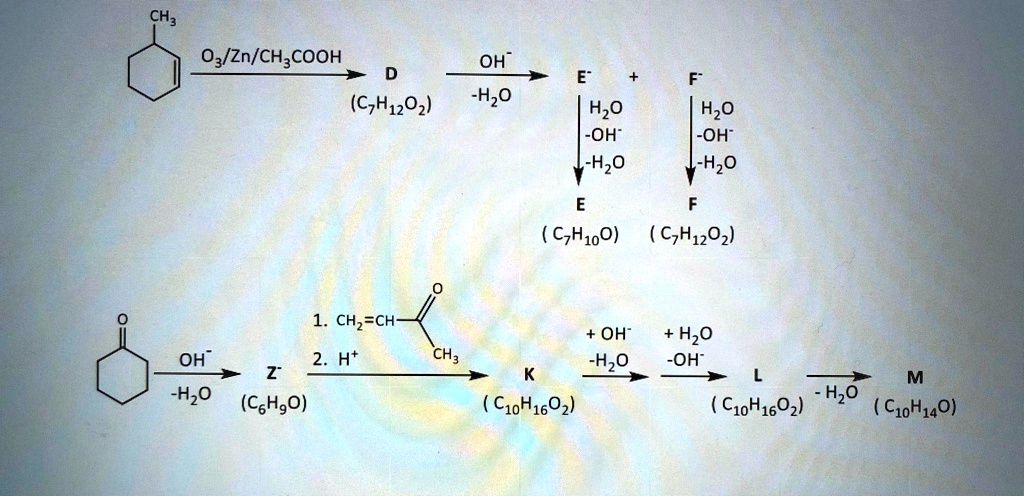
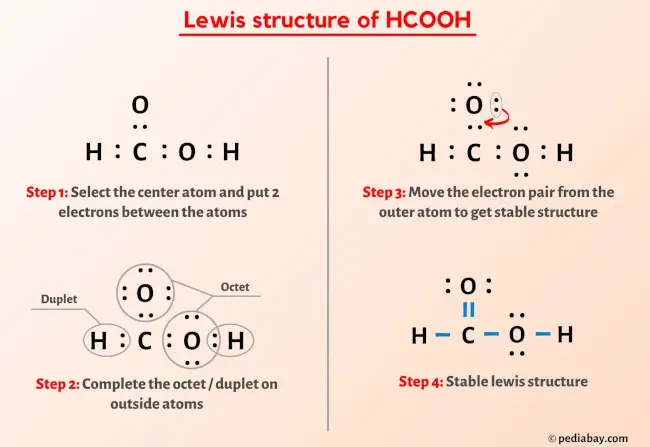
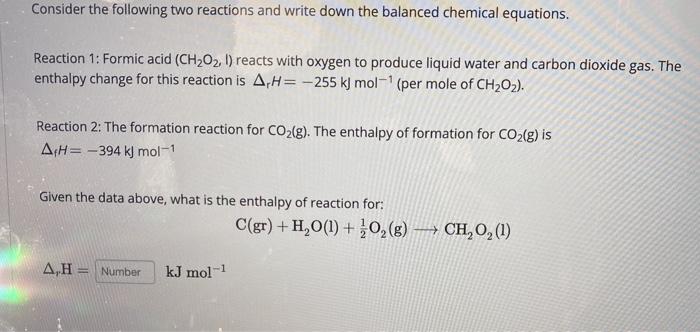Cyclohexanol Production via Esterification of Cyclohexene with Formic Acid and Subsequent Hydration of the EsterReaction Kinetics | Industrial & Engineering Chemistry Research
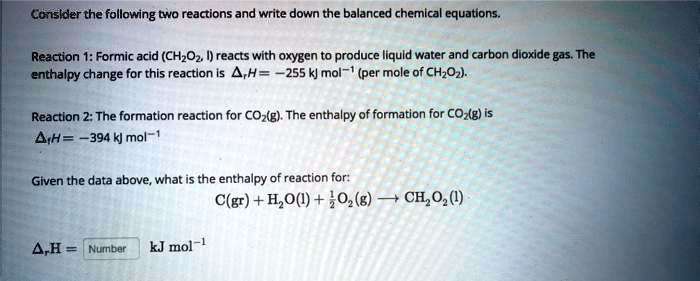
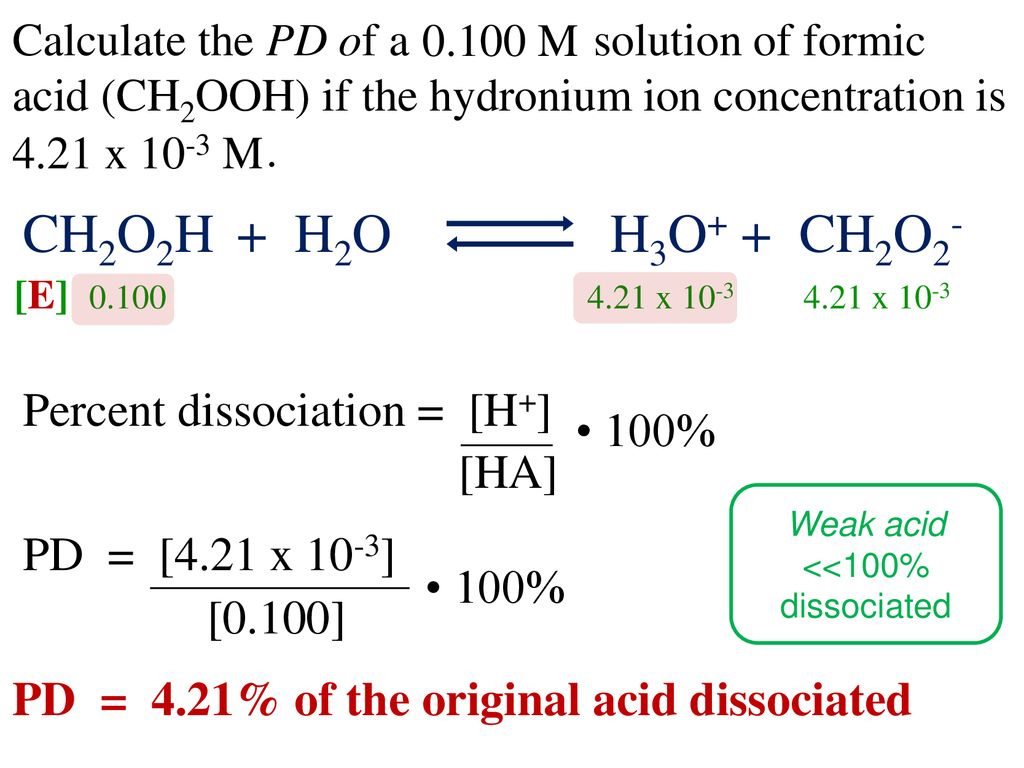
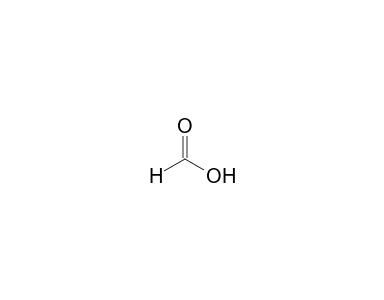
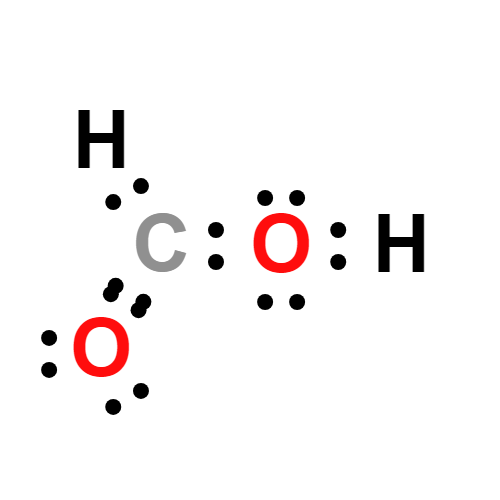
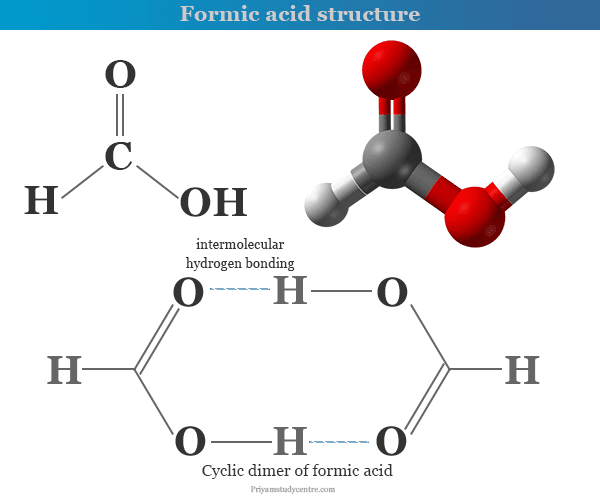
SOLVED: Consider the following two reactions and write down the balanced chemical equations: Reaction 1: Formic acid (CH2O2) reacts with oxygen to produce liquid water and carbon dioxide gas. The enthalpy change
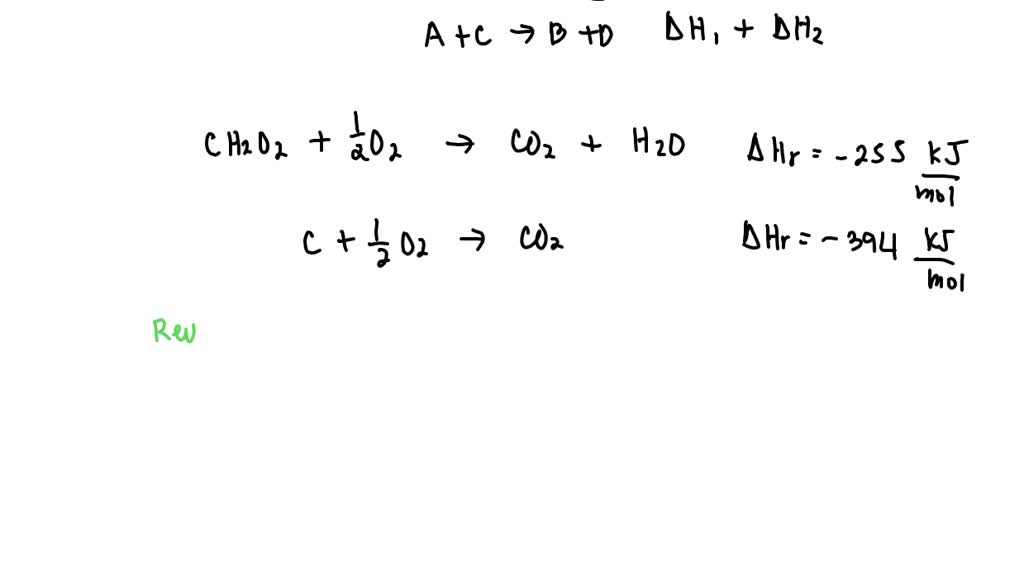
SOLVED: Consider the following two reactions and write down the balanced chemical equations: Reaction 1: Formic acid (CH2O2) reacts with oxygen to produce liquid water and carbon dioxide gas. The enthalpy change

Formic acid formation via direct hydration reaction (CO + H2O → HCOOH) on magnesia-silver composite - ScienceDirect