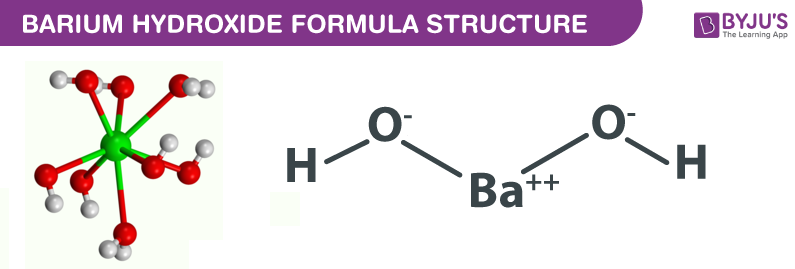
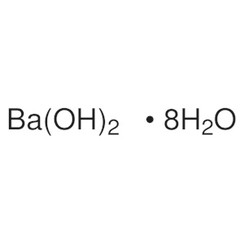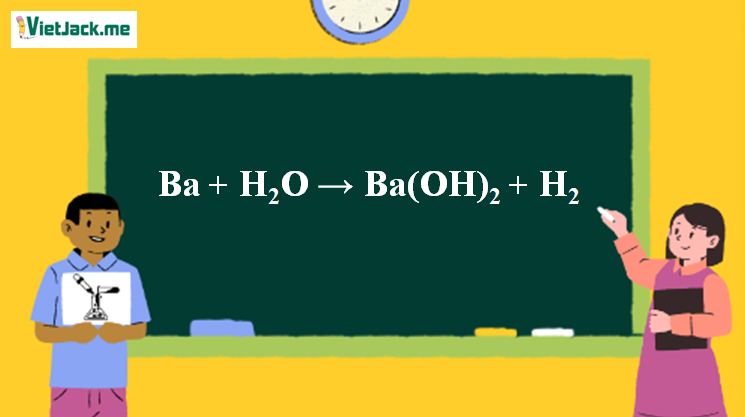SOLVED: Consider the acid-base reaction: 2 HNO2 (aq) + Ba(OH)2 (aq) â†' Ba(NO2)2 (aq) + 2 H2O Predict the products of the acid-base reaction: Ba2+ (aq) + 2 NO2- (aq) + 2
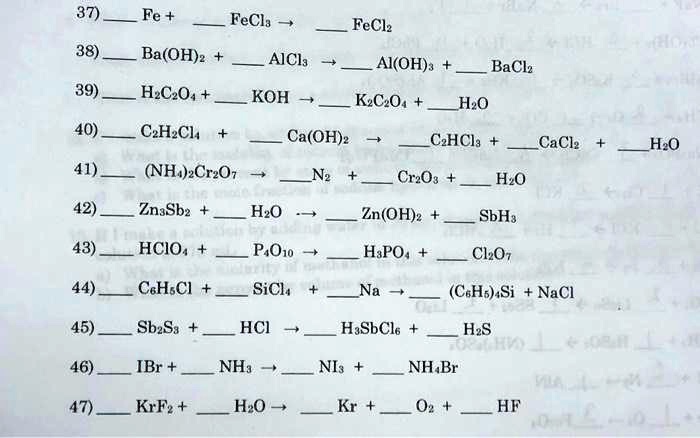
SOLVED: Texts: help please with this questions 37) Fe+ FeCl2 FeCl3 38) BaOH2 + AlCl3 -> Al(OH)3 + BaCl2 + H2O + CO2 + KOH + KCl + H2O + NH4Cl +

How to Write the Net Ionic Equation for Ba(OH)2 + H2SO4 = BaSO4 + H2O (Note: it should be 2H2O) - YouTube

Barium Hydroxide Monohydrate Ba (OH) 2. H2O - China Barium Hydroxide, Barium Hydroxide Monohydrate | Made-in-China.com

Figure 5 from Ba(OH)2 Equilibria in the System Ba-O-H-F, With Application to the Formation of Ba2YCu3O6.5 + x From BaF2-Precursors | Semantic Scholar

Ba + H2O =Ba(OH)2 +H2 Balanced Equation||Barium and Water=Barium hydroxide plus H2 Balanced Equation - YouTube